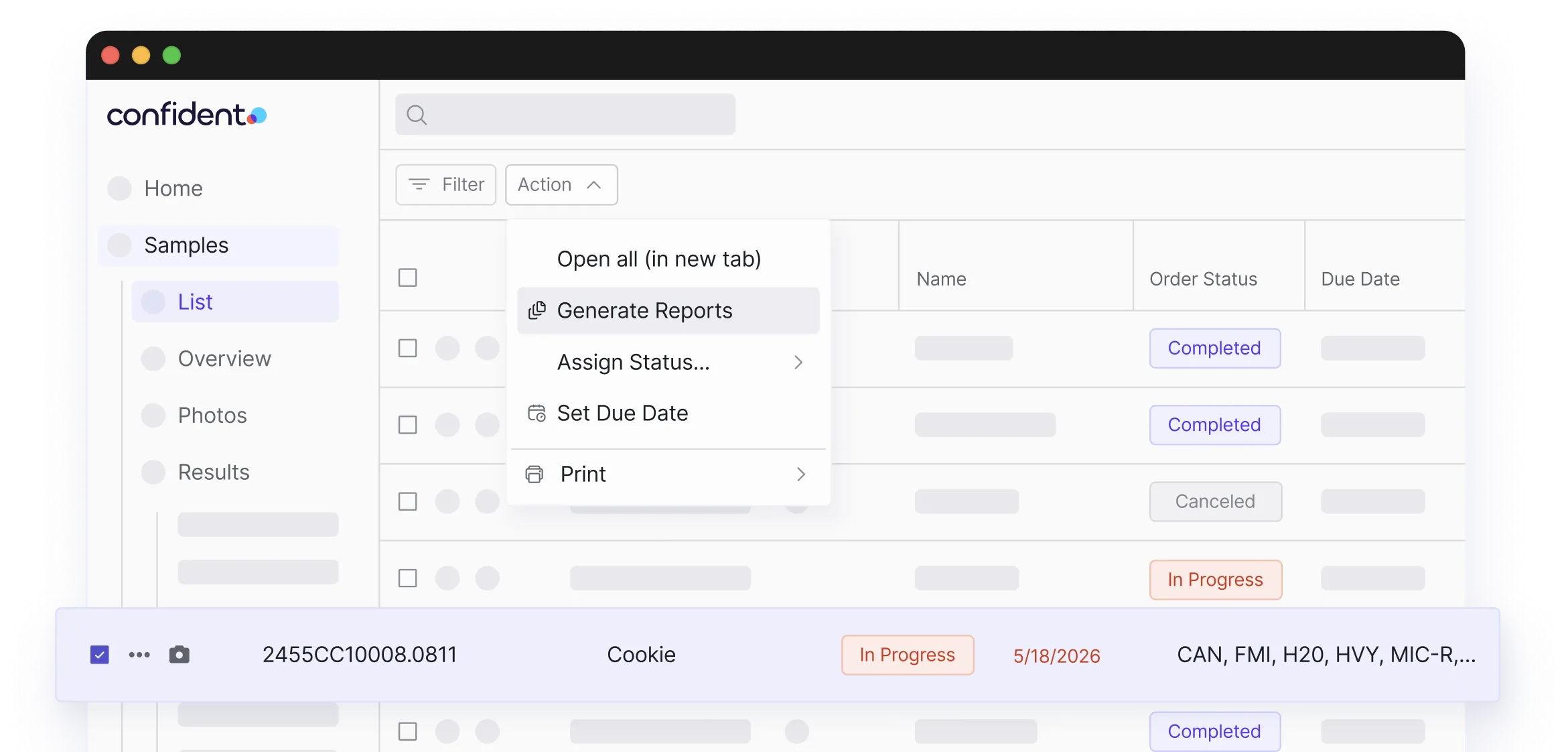
One Lab Account. Every Industry You Serve.
Serve every industry from one Confident LIMS account. Support for Ag, Cannabis, Environmental, Food, Nutraceuticals, Personal Care, Industrials, and Petrochem.
May 6, 2026
Announcements
From potency verification to contaminant screening, streamline your supplement testing workflows with automated compliance documentation and cGMP-ready reporting.


Automate potency, identity, and purity workflows from intake to certificate of analysis

Audit trails for cGMP, FDA DSHEA, and 21 CFR Part 111 — inspection-ready every day

Give supplement brands real-time access to potency results, COAs, and batch status

Quality Verification

Quality testing to verify product
specifications

Screening and quantification
for harmful substances